Announcement: Chapter quizzes are not working as of summer 2023.
![By John Kees (Own work) [<a href="http://creativecommons.org/licenses/by-sa/3.0">CC BY-SA 3.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AGrand_Canyon_Beauty.jpg">via Wikimedia Commons</a> The canyon is shown with many layers](https://opengeology.org/textbook/wp-content/uploads/2016/07/Grand_Canyon_Beauty.jpg)
- Explain the difference between relative time and numeric time
- Describe the five principles of stratigraphy
- Apply relative dating principles to a block diagram and interpret the sequence of geologic events
- Define an isotope, and explain alpha decay, beta decay, and electron capture as mechanisms of radioactive decay
- Describe how radioisotopic dating is accomplished and list the four key isotopes used
- Explain how carbon-14 forms in the atmosphere and how it is used in dating recent events
- Explain how scientists know the numeric age of the Earth and other events in Earth’s history
- Explain how sedimentary sequences can be dated using radioisotopes and other techniques
- Define a fossil and describe types of fossil preservation
- Outline how natural selection takes place as a mechanism of evolution
- Describe stratigraphic correlation
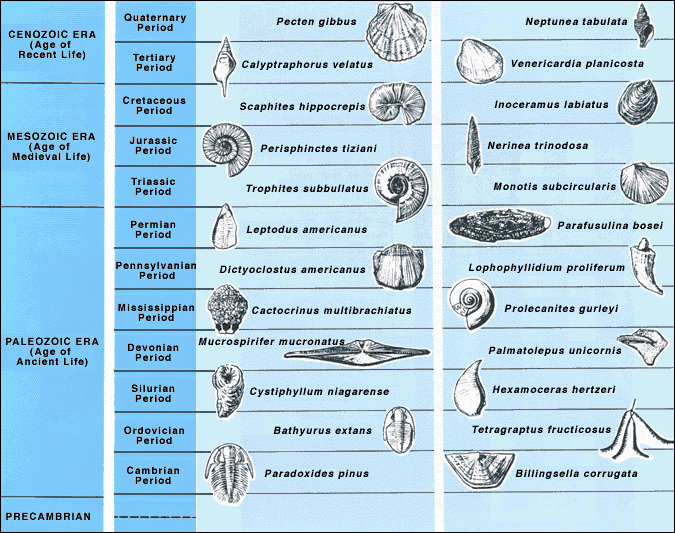
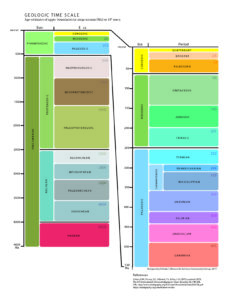
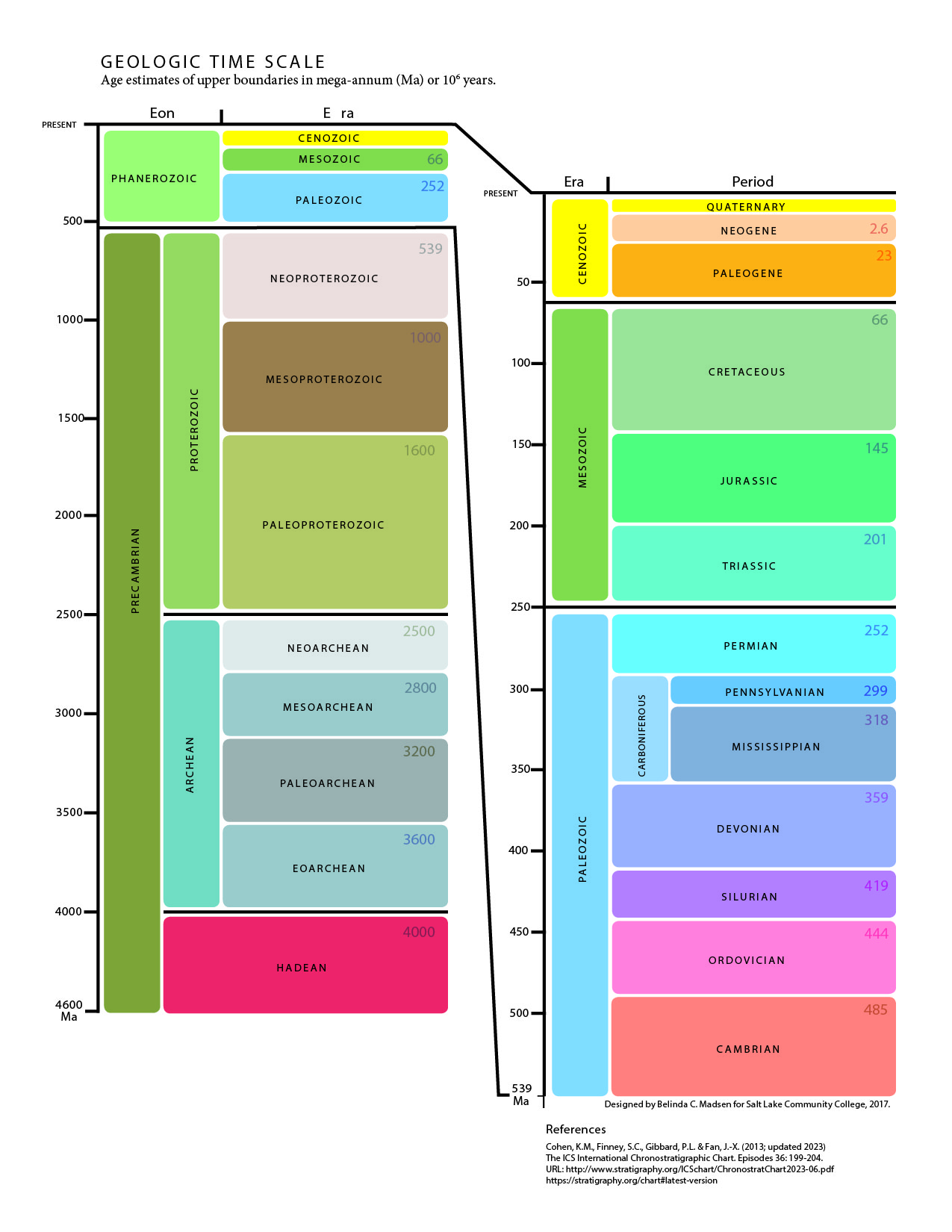
- List the eons, eras, and periods of the geologic time scale and explain the purpose behind the divisions
- Explain the relationship between time units and corresponding rock units—chronostratigraphy versus lithostratigraphy

The geologic time scale and basic outline of Earth’s history were worked out long before we had any scientific means of assigning numerical age units, like years, to events of Earth’s history. Working out Earth’s history depended on realizing some key principles of relative time. Nicolas Steno (1638-1686) introduced the basic principles of stratigraphy, the study of layered rocks, in 1669. William Smith (1769-1839), working with the strata of English coal mines, noticed that strata and their sequence were consistent throughout the region. Eventually, he produced the first national geologic map of Britain, becoming known as “the Father of English Geology.” Nineteenth-century scientists developed a relative time scale using Steno’s principles, with names derived from the characteristics of the rocks in those areas. The figure of this geologic time scale shows the names of the units and subunits. Using this time scale, geologists can place all events of Earth’s history in order without ever knowing their numerical ages. The specific events within Earth’s history are discussed in Chapter 8.
7.1 Relative Dating
Relative dating is the process of determining if one rock or geologic event is older or younger than another, without knowing their specific ages—i.e., how many years ago the object was formed. The principles of relative time are simple, even obvious now, but were not generally accepted by scholars until the scientific revolution of the 17th and 18th centuries. James Hutton (see Chapter 1) realized geologic processes are slow and his ideas on uniformitarianism (i.e., “the present is the key to the past”) provided a basis for interpreting rocks of the Earth using scientific principles.
7.1.1 Relative Dating Principles
Stratigraphy is the study of layered sedimentary rocks. This section discusses principles of relative time used in all of geology, but are especially useful in stratigraphy.
![By Wilson44691 (Own work) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3AIsfjordenSuperposition.jpg">via Wikimedia Commons</a> Photo of superposed strata with the younger on top of the older](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.2_IsfjordenSuperposition-228x300.jpg)
Principle of Original Horizontality: Layers of rocks deposited from above, such as sediments and lava flows, are originally laid down horizontally. The exception to this principle is at the margins of basins, where the strata can slope slightly downward into the basin.

Principle of Lateral Continuity: Within the depositional basin, strata are continuous in all directions until they thin out at the edge of that basin. Of course, all strata eventually end, either by hitting a geographic barrier, such as a ridge, or when the depositional process extends too far from its source, either a sediment source or a volcano. Strata that are cut by a canyon later remain continuous on either side of the canyon.
![By Thomas Eliasson of Geological Survey of Sweden http://www.flickr.com/people/geologicalsurveyofsweden/ [<a href="http://creativecommons.org/licenses/by/2.0">CC BY 2.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AMultiple_Igneous_Intrusion_Phases_Kosterhavet_Sweden.jpg">via Wikimedia Commons</a> Photo of rock outcrop with a dike cutting through an older rock and another dike cutting across that one.](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.6_Multiple_Igneous_Intrusion_Phases_Kosterhavet_Sweden-225x300.jpg)
Principle of Inclusions: When one rock formation contains pieces or inclusions of another rock, the included rock is older than the host rock.
Principle of Fossil Succession: Evolution has produced a succession of unique fossils that correlate to the units of the geologic time scale. Assemblages of fossils contained in strata are unique to the time they lived and can be used to correlate rocks of the same age across a wide geographic distribution. Assemblages of fossils refer to groups of several unique fossils occurring together.
7.1.2 Grand Canyon Example

The Grand Canyon of Arizona illustrates the stratigraphic principles. The photo shows layers of rock on top of one another in order, from the oldest at the bottom to the youngest at the top, based on the principle of superposition. The predominant white layer just below the canyon rim is the Coconino Sandstone. This layer is laterally continuous, even though the intervening canyon separates its outcrops. The rock layers exhibit the principle of lateral continuity, as they are found on both sides of the Grand Canyon which has been carved by the Colorado River.
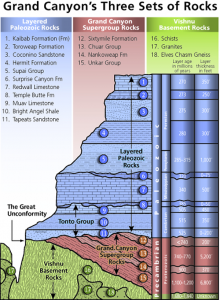
The diagram called “Grand Canyon’s Three Sets of Rocks” shows a cross-section of the rocks exposed on the walls of the Grand Canyon, illustrating the principle of cross-cutting relationships, superposition, and original horizontality. In the lowest parts of the Grand Canyon are the oldest sedimentary formations, with igneous and metamorphic rocks at the bottom. The principle of cross-cutting relationships shows the sequence of these events. The metamorphic schist (#16) is the oldest rock formation and the cross-cutting granite intrusion (#17) is younger. As seen in the figure, the other layers on the walls of the Grand Canyon are numbered in reverse order with #15 being the oldest and #1 the youngest. This illustrates the principle of superposition. The Grand Canyon region lies in the Colorado Plateau, which is characterized by horizontal or nearly horizontal strata, which follow the principle of original horizontality. These rock strata have been barely disturbed from their original deposition, except by a broad regional uplift.
![By Simeon87 (Own work) [<a href="http://creativecommons.org/licenses/by-sa/3.0">CC BY-SA 3.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AGrand_Canyon_with_Snow_4.JPG">via Wikimedia Commons</a> The red rocks are layered, the dark rocks are not.](https://opengeology.org/textbook/wp-content/uploads/2016/07/Grand_Canyon_Nonconformity-300x225.jpg)
7.1.3 Unconformities

There are three types of unconformities, nonconformity, disconformity, and angular unconformity. A nonconformity occurs when sedimentary rock is deposited on top of igneous and metamorphic rocks as is the case with the contact between the strata and basement rocks at the bottom of the Grand Canyon.
The strata in the Grand Canyon represent alternating marine transgressions and regressions where sea level rose and fell over millions of years. When the sea level was high marine strata formed. When sea-level fell, the land was exposed to erosion creating an unconformity. In the Grand Canyon cross-section, this erosion is shown as heavy wavy lines between the various numbered strata. This is a type of unconformity called a disconformity, where either non-deposition or erosion took place. In other words, layers of rock that could have been present, are absent. The time that could have been represented by such layers is instead represented by the disconformity. Disconformities are unconformities that occur between parallel layers of strata indicating either a period of no deposition or erosion.
![By Doug Dolde / Doug Dolde at en.wikipedia (Contax 645, 140mm, Leaf Aptus 75S) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3AView_from_Lipan_Point.jpg">via Wikimedia Commons</a> The rocks are mostly red.](https://opengeology.org/textbook/wp-content/uploads/2016/07/View_from_Lipan_Point-300x224.jpg)
Here are three graphical illustrations of the three types of unconformity.

Disconformity, where is a break or stratigraphic absence between strata in an otherwise parallel sequence of strata.
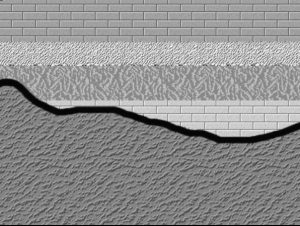
Nonconformity, where sedimentary strata are deposited on crystalline (igneous or metamorphic) rocks.
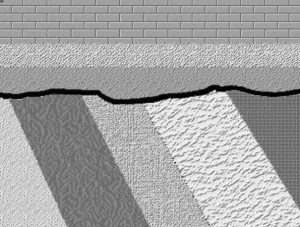
Angular unconformity, where sedimentary strata are deposited on a terrain developed on sedimentary strata that have been deformed by tilting, folding, and/or faulting. so that they are no longer horizontal.
7.1.3 Applying Relative Dating Principles
![By Woudloper (Own work) [<a href="http://creativecommons.org/licenses/by-sa/1.0">CC BY-SA 1.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3ACross-cutting_relations.svg">via Wikimedia Commons</a>](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.4_Block_diagram-300x159.jpg)
[ays_quiz id=”40″]
7.2 Absolute Dating

Relative time allows scientists to tell the story of Earth’s events but does not provide specific numeric ages, and thus, the rate at which geologic processes operate. Based on Hutton’s principle of uniformitarianism (see Chapter 1), early geologists surmised geological processes work slowly and the Earth is very old. Relative dating principles were how scientists interpreted Earth’s history until the end of the 19th Century. Because science advances as technology advances, the discovery of radioactivity in the late 1800s provided scientists with a new scientific tool called radioisotopic dating. Using this new technology, they could assign specific time units, in this case years, to mineral grains within a rock. These numerical values are not dependent on comparisons with other rocks such as with relative dating, so this dating method is called absolute dating. There are several types of absolute dating discussed in this section but radioisotopic dating is the most common and therefore is the focus on this section.
7.2.1 Radioactive Decay
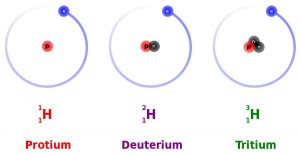
All elements on the Periodic Table of Elements (see Chapter 3) contain isotopes. An isotope is an atom of an element with a different number of neutrons. For example, hydrogen (H) always has 1 proton in its nucleus (the atomic number), but the number of neutrons can vary among the isotopes (0, 1, 2). Recall that the number of neutrons added to the atomic number gives the atomic mass. When hydrogen has 1 proton and 0 neutrons it is sometimes called protium (1H), when hydrogen has 1 proton and 1 neutron it is called deuterium (2H), and when hydrogen has 1 proton and 2 neutrons it is called tritium (3H).
Many elements have both stable and unstable isotopes. For the hydrogen example, 1H and 2H are stable, but 3H is unstable. Unstable isotopes, called radioactive isotopes, spontaneously decay over time releasing subatomic particles or energy in a process called radioactive decay. When this occurs, an unstable isotope becomes a more stable isotope of another element. For example, carbon-14 (14C) decays to nitrogen-14 (14N).
![By Sbyrnes321 (Own work) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3AHalflife-sim.gif">via Wikimedia Commons</a> The many looks more smooth](https://opengeology.org/textbook/wp-content/uploads/2016/07/Halflife-sim.gif)
The principles behind this dating method require two key assumptions. First, the mineral grains containing the isotope formed at the same time as the rock, such as minerals in an igneous rock that crystallized from magma. Second, the mineral crystals remain a closed system, meaning they are not subsequently altered by elements moving in or out of them.

These requirements place some constraints on the kinds of rock suitable for dating, with igneous rock being the best. Metamorphic rocks are crystalline, but the processes of metamorphism may reset the clock and derived ages may represent a smear of different metamorphic events rather than the age of original crystallization. Detrital sedimentary rocks contain clasts from separate parent rocks from unknown locations and derived ages are thus meaningless. However, sedimentary rocks with precipitated minerals, such as evaporites, may contain elements suitable for radioisotopic dating. Igneous pyroclastic layers and lava flows within a sedimentary sequence can be used to date the sequence. Cross-cutting igneous rocks and sills can be used to bracket the ages of affected, older sedimentary rocks. The resistant mineral zircon, found as clasts in many ancient sedimentary rocks, has been successfully used for establishing very old dates, including the age of Earth’s oldest known rocks. Knowing that zircon minerals in metamorphosed sediments came from older rocks that are no longer available for study, scientists can date zircon to establish the age of the pre-metamorphic source rocks.
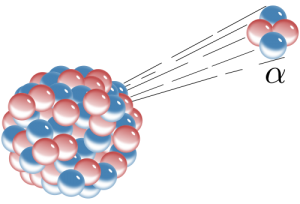
There are several ways radioactive atoms decay. We will consider three of them here—alpha decay, beta decay, and electron capture. Alpha decay is when an alpha particle, which consists of two protons and two neutrons, is emitted from the nucleus of an atom. This also happens to be the nucleus of a helium atom; helium gas may get trapped in the crystal lattice of a mineral in which alpha decay has taken place. When an atom loses two protons from its nucleus, lowering its atomic number, it is transformed into an element that is two atomic numbers lower on the Periodic Table of the Elements.
![By Sandbh (Own work) [<a href="http://creativecommons.org/licenses/by-sa/4.0">CC BY-SA 4.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3APeriodic_Table_Chart.png">via Wikimedia Commons</a> Simplified Periodic Table of the Elements](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.11_periodic_table-300x160.png)
B
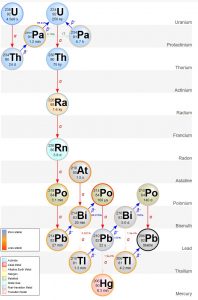
Beta decay is when a neutron in its nucleus splits into an electron and a proton. The electron is emitted from the nucleus as a beta ray. The new proton increases the element’s atomic number by one, forming a new element with the same atomic mass as the parent isotope. For example, 234Th is unstable and undergoes beta decay to form protactinium-234 (234Pa), which also undergoes beta decay to form uranium-234 (234U). Notice these are all isotopes of different elements but they have the same atomic mass of 234. The decay process of radioactive elements like uranium keeps producing radioactive parents and daughters until a stable, or non-radioactive, daughter is formed. Such a series is called a decay chain. The decay chain of the radioactive parent isotope 238U progresses through a series of alpha (red arrows on the adjacent figure) and beta decays (blue arrows) until it forms the stable daughter isotope, lead-206 (206Pb).
![By Pamputt (Own work) [<a href="http://creativecommons.org/licenses/by-sa/4.0">CC BY-SA 4.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AAtomic_rearrangement_following_an_electron_capture.svg">via Wikimedia Commons</a> It shows two paths](https://opengeology.org/textbook/wp-content/uploads/2016/07/Atomic_rearrangement_following_an_electron_capture.svg_-254x300.png)
| Elements | Parent symbol | Daughter symbol | Half-life |
| Uranium-238/Lead-206 | 238U | 206Pb | 4.5 billion years |
| Uranium-235/Lead-207 | 235U | 207Pb | 704 million years |
| Potassium-40/Argon-40 | 40K | 40Ar | 1.25 billion years |
| Rubidium-87/Strontium-87 | 87Rb | 87Sr | 48.8 billion years |
| Carbon-14/Nitrogen-14 | 14C | 14N | 5,730 years |
Some common isotopes used for radioisotopic dating.
7.2.2 Radioisotopic Dating

For a given sample of rock, how is the dating procedure carried out? The parent and daughter isotopes are separated out of the mineral using chemical extraction. In the case of uranium, 238U and 235U isotopes are separated out together, as is the 206Pb and 207Pb with an instrument called a mass spectrometer.
![By Krishnavedala (Own work) [<a href="http://creativecommons.org/publicdomain/zero/1.0/deed.en">CC0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AHalf_times.svg">via Wikimedia Commons</a> The graph gets progressively taller](https://opengeology.org/textbook/wp-content/uploads/2016/07/Half_times.svg_-300x300.png)
| Half-lives
(#) |
Parent present (%) | Daughter present
(%) |
Daughter/
Parent ratio |
Parent/
Daughter ratio |
| Start the clock | 100 | 0 | 0 | infinite |
| 1 | 50 | 50 | 1 | 1 |
| 2 | 25 | 75 | 3 | 0.33 |
| 3 | 12.5 | 87.5 | 7 | 0.143 |
| 4 | 6.25 | 93.75 | 15 | 0.0667 |
| 5 | 3.125 | 96.875 | 31 | 0.0325 |
| 10 | 0.098 | 99.9 | 1023 | 0.00098 |
Ratio of parent to daughter in terms of half-life.
![By Mike Christie (Own work) [<a href="http://creativecommons.org/licenses/by-sa/3.0">CC BY-SA 3.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AAccelerator_mass_spectrometer_schematic_for_radiocarbon.svg">via Wikimedia Commons</a> It shows the isotopes separating](https://opengeology.org/textbook/wp-content/uploads/2016/07/Accelerator_mass_spectrometer_schematic_for_radiocarbon.svg_-300x111.png)
Radiocarbon dating relies on daughter-to-parent ratios derived from a known quantity of parent 14C. Early applications of carbon dating assumed the production and concentration of 14C in the atmosphere remained fairly constant for the last 50,000 years. However, it is now known that the amount of parent 14C levels in the atmosphere has varied. Comparisons of carbon ages with tree-ring data and other data for known events have allowed reliable calibration of the radiocarbon dating method. Taking into account carbon-14 baseline levels must be calibrated against other reliable dating methods, carbon dating has been shown to be a reliable method for dating archaeological specimens and very recent geologic events.
7.2.3 Age of the Earth
![By Tim Bertelink (Own work) [<a href="http://creativecommons.org/licenses/by-sa/4.0">CC BY-SA 4.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AHadean.png">via Wikimedia Commons</a> The surface of Earth is full of volcanoes.](https://opengeology.org/textbook/wp-content/uploads/2017/03/Hadean-300x161.png)
In the 1950’s, Clair Patterson (1922–1995) thought he could determine the age of the Earth using radioactive isotopes from meteorites, which he considered to be early solar system remnants that were present at the time Earth was forming. Patterson analyzed meteorite samples for uranium and lead using a mass spectrometer. He used the uranium/lead dating technique in determining the age of the Earth to be 4.55 billion years, give or take about 70 million (± 1.5%). The current estimate for the age of the Earth is 4.54 billion years, give or take 50 million (± 1.1%). It is remarkable that Patterson, who was still a graduate student at the University of Chicago, came up with a result that has been little altered in over 60 years, even as technology has improved dating methods.
7.2.4 Dating Geological Events

Radioactive isotopes of elements that are common in mineral crystals are useful for radioisotopic dating. The uranium/lead method, with its two cross-checking clocks, is most often used with crystals of the mineral zircon (ZrSiO4) where uranium can substitute for zirconium in the crystal lattice. Zircon is resistant to weathering which makes it useful for dating geological events in ancient rocks. During metamorphic events, zircon crystals may form multiple crystal layers, with each layer recording the isotopic age of an event, thus tracing the progress of several metamorphic events.
Geologists have used zircon grains to do some amazing studies that illustrate how scientific conclusions can change with technological advancements. Zircon crystals from Western Australia that formed when the crust first differentiated from the mantle 4.4 billion years ago have been determined to be the oldest known rocks. The zircon grains were incorporated into metasedimentary host rocks, sedimentary rocks showing signs of having undergone partial metamorphism. The host rocks were not very old but the embedded zircon grains were created 4.4 billion years ago and survived the subsequent processes of weathering, erosion, deposition, and metamorphism. From other properties of the zircon crystals, researchers concluded that not only were continental rocks exposed above sea level, but also that conditions on the early Earth were cool enough for liquid water to exist on the surface. The presence of liquid water allowed the processes of weathering and erosion to take place. Researchers at UCLA studied 4.1 billion-year-old zircon crystals and found carbon in the zircon crystals that may be biogenic in origin, meaning that life may have existed on Earth much earlier than previously thought.
Igneous rocks are best suited for radioisotopic dating because their primary minerals provide dates of crystallization from magma. Metamorphic processes tend to reset the clocks and smear the igneous rock’s original date. Detrital sedimentary rocks are less useful because they are made of minerals derived from multiple parent sources with potentially many dates. However, scientists can use igneous events to date sedimentary sequences. For example, if sedimentary strata are between a lava flow and volcanic ash bed with radioisotopic dates of 54 million years and 50 million years, then geologists know the sedimentary strata and its fossils formed between 54 and 50 million years ago. Another example would be a 65 million-year-old volcanic dike that cut across sedimentary strata. This provides an upper limit age on the sedimentary strata, so these strata would be older than 65 million years. Potassium is common in evaporite sediments and has been used for potassium/argon dating. Primary sedimentary minerals containing radioactive isotopes like 40K, have provided dates for important geologic events.
7.2.5 Other Absolute Dating Techniques
![By Zkeizars (Own work) [<a href="http://www.gnu.org/copyleft/fdl.html">GFDL</a> or <a href="http://creativecommons.org/licenses/by-sa/4.0-3.0-2.5-2.0-1.0">CC BY-SA 4.0-3.0-2.5-2.0-1.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AKeizars_TLexplained2.jpg">via Wikimedia Commons</a> The diagram explains the details of the technique, showing trapped electrons.](https://opengeology.org/textbook/wp-content/uploads/2016/07/Keizars_TLexplained2-300x247.jpg)
![Rob Lavinsky, <a rel="nofollow" class="external text" href="http://www.irocks.com/">iRocks.com</a> – CC-BY-SA-3.0 [<a href="http://creativecommons.org/licenses/by-sa/3.0">CC BY-SA 3.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AApatite-(CaF)-280343.jpg">via Wikimedia Commons</a> The crystal is hexagonal and light green.](https://opengeology.org/textbook/wp-content/uploads/2017/01/Apatite-CaF-280343-300x267.jpg)
[ays_quiz id=”41″]
7.3 Fossils and Evolution
![By H. Raab (User: Vesta) (Own work) [<a href="http://creativecommons.org/licenses/by-sa/3.0">CC BY-SA 3.0</a> or <a href="http://www.gnu.org/copyleft/fdl.html">GFDL</a>], <a href="https://commons.wikimedia.org/wiki/File%3AArchaeopteryx_lithographica_(Berlin_specimen).jpg">via Wikimedia Commons</a> Image of the Archaeopteryx fossil that show features of both reptiles and birds. This is a famous transition fossil between reptiles and birds.](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.3_Archaeopteryx_lithographica-222x300.jpg)
7.3.1 Types of Preservation
![By Wilson44691 (Own work) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3AElrathiakingiUtahWheelerCambrian.jpg">via Wikimedia Commons</a> Image of three lobes of an external skeleton like an insect has today.](https://opengeology.org/textbook/wp-content/uploads/2016/07/ElrathiakingiUtahWheelerCambrian-243x300.jpg)
Actual preservation is a rare form of fossilization where the original materials or hard parts of the organism are preserved. Preservation of soft tissue is very rare since these organic materials easily disappear because of bacterial decay. Examples of actual preservation are unaltered biological materials like insects in amber or original minerals like mother-of-pearl on the interior of a shell. Another example is mammoth skin and hair preserved in post-glacial deposits in the Arctic regions. Rare mummification has left fragments of soft tissue, skin, and sometimes even blood vessels of dinosaurs, from which proteins have been isolated and evidence for DNA fragments have been discovered.
Molds and casts form when the original material of the organism dissolves and leaves a cavity in the surrounding rock. The shape of this cavity is an external mold. If the mold is subsequently filled with sediments or a mineral precipitate, the organism’s external shape is preserved as a cast. Sometimes internal cavities of organisms, such as internal casts of clams, snails, and even skulls are preserved as internal casts showing details of soft structures. If the chemistry is right, and burial is rapid, mineral nodules form around soft structures preserving the three-dimensional detail. This is called authigenic mineralization.

Trace fossils are indirect evidence left behind by an organism, such as burrows and footprints, as it lived its life. Ichnology is specifically the study of prehistoric animal tracks. Dinosaur tracks testify to their presence and movement over an area, and even provide information about their size, gait, speed, and behavior. Burrows dug by tunneling organisms tell of their presence and mode of life. Other trace fossils include fossilized feces called coprolites and stomach stones called gastroliths that provide information about diet and habitat.
7.3.2 Evolution
Evolution has created a variety of ancient fossils that are important to stratigraphic correlation. (See Chapter 7 and Chapter 5) This section is a brief discussion of the process of evolution. The British naturalist Charles Darwin (1809-1882) recognized that life forms evolve into progeny life forms. He proposed natural selection—which operated on organisms living under environmental conditions that posed challenges to survival—was the mechanism driving the process of evolution forward.
The basic classification unit of life is the species: a population of organisms that exhibit shared characteristics and are capable of reproducing fertile offspring. For a species to survive, each individual within a particular population is faced with challenges posed by the environment and must survive them long enough to reproduce. Within the natural variations present in the population, there may be individuals possessing characteristics that give them some advantage in facing environmental challenges. These individuals are more likely to reproduce and pass these favored characteristics on to successive generations. If sufficient individuals in a population fail to surmount the challenges of the environment and the population cannot produce enough viable offspring, the species becomes extinct. The average lifespan of a species in the fossil record is around a million years. That life still exists on Earth shows the role and importance of evolution as a natural process in meeting the continual challenges posed by our dynamic Earth. If the inheritance of certain distinctive characteristics is sufficiently favored over time, populations may become genetically isolated from one another, eventually resulting in the evolution of separate species. This genetic isolation may also be caused by a geographic barrier, such as an island surrounded by ocean. This theory of evolution by natural selection was elaborated by Darwin in his book On the Origin of Species (see Chapter 1). Since Darwin’s original ideas, technology has provided many tools and mechanisms to study how evolution and speciation take place and this arsenal of tools is growing. Evolution is well beyond the hypothesis stage and is a well-established theory of modern science.
Variation within populations occurs by the natural mixing of genes through sexual reproduction or from naturally occurring mutations. Some of this genetic variation can introduce advantageous characteristics that increase the individual’s chances of survival. While some species in the fossil record show little morphological change over time, others show gradual or punctuated changes, within which intermediate forms can be seen.
[ays_quiz id=”42″]
7.4 Correlation
![By Osvaldocangaspadilla (Own work) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3ASnider-Pellegrini_Wegener_fossil_map.svg">via Wikimedia Commons</a> There are four different fossil organisms that connect South America, Africa, India, Antartica, and Australia.](https://opengeology.org/textbook/wp-content/uploads/2017/03/Wegener_fossil_map.svg_-300x231.png)
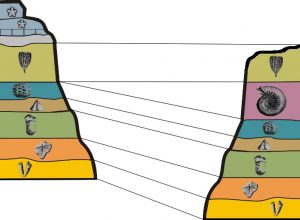
7.4.1 Stratigraphic Correlation
Stratigraphic correlation is the process of establishing which sedimentary strata are the same age at distant geographical areas by means of their stratigraphic relationship. Geologists construct geologic histories of areas by mapping and making stratigraphic columns which are detailed descriptions of the strata from bottom to top. An example of stratigraphic relationships and correlation between Canyonlands National Park and Zion National Park in Utah. At Canyonlands, the Navajo Sandstone overlies the Kayenta Formation which overlies the cliff-forming Wingate Formation. In Zion, the Navajo Sandstone overlies the Kayenta Formation which overlies the cliff-forming Moenave Formation. Based on the stratigraphic relationship, the Wingate and Moenave Formations correlate. These two formations have unique names because their composition and outcrop pattern is slightly different. Other strata in the Colorado Plateau and their sequence can be recognized and correlated over thousands of square miles.
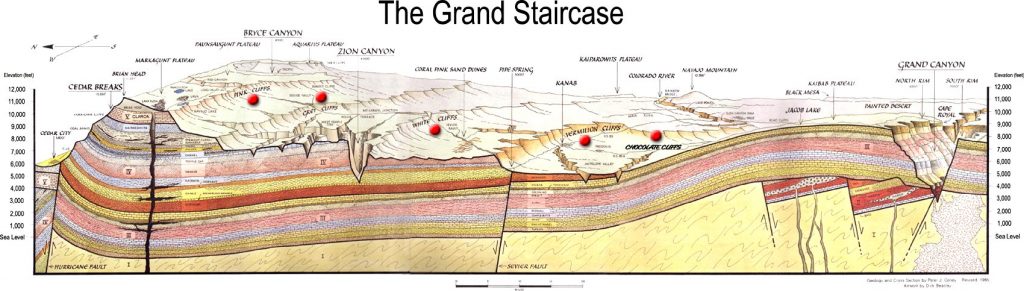
7.4.2 Lithostratigraphic Correlation
![By Diliff (taken by Diliff) [<a href="http://www.gnu.org/copyleft/fdl.html">GFDL</a>, <a href="http://creativecommons.org/licenses/by-sa/3.0/">CC-BY-SA-3.0</a> or <a href="http://creativecommons.org/licenses/by-sa/2.5-2.0-1.0">CC BY-SA 2.5-2.0-1.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AZion_angels_landing_view.jpg">via Wikimedia Commons</a> View of Navajo Sandstone from Angel's Landing in Zion National Park](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.28_Navajo_Sandstone_Zion_angels_landing_view-293x300.jpg)
![By G. Thomas at English Wikipedia [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3AStevensArchUT.jpg">via Wikimedia Commons</a> Stevens Arch in the Navajo Sandstone at Coyote Gulch some 125 miles away from Zion National Park](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.29_Navajo_Sandstone_StevensArchUT-300x212.jpg)
7.4.3 Chronostratigraphic Correlation
Chronostratigraphic correlation matches rocks of the same age, even though they are made of different lithologies. Different lithologies of sedimentary rocks can form at the same time at different geographic locations because depositional environments vary geographically. For example, at any one time in a marine setting, there could be this sequence of depositional environments from the beach to deep marine: beach, near-shore area, shallow marine lagoon, reef, slope, and deep marine. Each depositional environment will have a unique sedimentary rock formation. On the figure of the Permian Capitan Reef at Guadalupe National Monument in West Texas, the red line shows a chronostratigraphic timeline that represents a snapshot in time. Shallow-water marine lagoon/back reef area is light blue, the main Capitan reef is dark blue, and deep-water marine siltstone is yellow. All three of these unique lithologies were forming at the same time in the Permian along this red timeline.
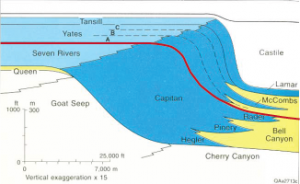
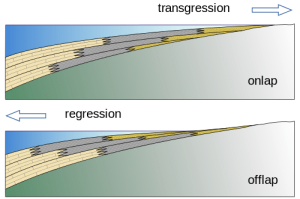
7.4.4 Biostratigraphic Correlation
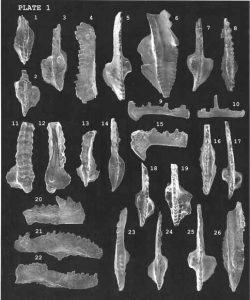
Biostratigraphic correlation uses index fossils to determine strata ages. Index fossils represent assemblages or groups of organisms that were uniquely present during specific intervals of geologic time. Assemblages refer to a group of fossils. Fossils allow geologists to assign a formation to an absolute date range, such as the Jurassic Period (199 to 145 million years ago), rather than a relative time scale. In fact, most of the geologic time ranges are mapped to fossil assemblages. The most useful index fossils come from lifeforms that were geographically widespread and had a species lifespan that was limited to a narrow time interval. In other words, index fossils can be found in many places around the world, but only during a narrow time frame. Some of the best fossils for biostratigraphic correlation are microfossils, most of which came from single-celled organisms. As with microscopic organisms today, they are widely distributed across many environments throughout the world. Some of these microscopic organisms had hard parts, such as exoskeletons or outer shells, making them better candidates for preservation. Foraminifera, single-celled organisms with calcareous shells, are an example of an especially useful index fossil for the Cretaceous Period and Cenozoic Era.
Conodonts are another example of microfossils useful for biostratigraphic correlation of the Cambrian through Triassic Periods. Conodonts are tooth-like phosphatic structures of an eel-like multi-celled organism that had no other preservable hard parts. The conodont-bearing creatures lived in shallow marine environments all over the world. Upon death, the phosphatic hard parts were scattered into the rest of the marine sediments. These distinctive tooth-like structures are easily collected and separated from limestone in the laboratory.
![By Philippe Janvier, 1997 (Tree of Life Web Project) [<a href="http://creativecommons.org/licenses/by/3.0">CC BY 3.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3AEuconodonta.gif">via Wikimedia Commons</a> Artists rendering of what the conodont anmal might have looked like, an eel-like creature with large eyes and an apparatus of conodonts as mouthparts.](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.33_Euconodonta.gif)
7.4.5 Geologic Time Scale
![<code>By WoudloperDerivative work: Hardwigg (File:Geologic_clock.jpg) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3AGeologic_Clock_with_events_and_periods.svg">via Wikimedia Commons</a></code> The circle starts at 4.6 billion years ago, then loops around to zero.](https://opengeology.org/textbook/wp-content/uploads/2017/03/GeologicClock-300x288.png)
The geologic time scale was developed during the 19th century using the principles of stratigraphy. The relative order of the time units was determined before geologists had the tools to assign numerical ages to periods and events. Biostratigraphic correlation using fossils to assign era and period names to sedimentary rocks on a worldwide scale. With the expansion of science and technology, some geologists think the influence of humanity on natural processes has become so great they are suggesting a new geologic time period, known as the Anthropocene.
[ays_quiz id=”43″]
Chapter Summary
Events in Earth’s history can be placed in sequence using the five principles of relative dating. The geologic time scale was completely worked out in the 19th Century using these principles without knowing any actual numeric ages for the events. The discovery of radioactivity in the late 1800s enabled absolute dating, the assignment of numerical ages to events in the Earth’s history, using the decay of unstable radioactive isotopes. Accurately interpreting radioisotopic dating data depends on the type of rock tested and accurate assumptions about isotope baseline values. With a combination of relative and absolute dating, the history of geological events, the age of Earth, and a geologic time scale have been determined with considerable accuracy. Stratigraphic correlation is an additional tool used for understanding how depositional environments change geographically. Geologic time is vast, providing plenty of time for the evolution of various lifeforms, and some of these have become preserved as fossils that can be used for biostratigraphic correlation. The geologic time scale is continuous, although the rock record may be broken because rocks representing certain time periods may be missing.
[ays_quiz id=”44″]
References
- Allison, P.A., and Briggs, D.E.G., 1993, Exceptional fossil record: Distribution of soft-tissue preservation through the Phanerozoic: Geology, v. 21, no. 6, p. 527–530.
- Bell, E.A., Boehnke, P., Harrison, T.M., and Mao, W.L., 2015, Potentially biogenic carbon preserved in a 4.1 billion-year-old zircon: Proc. Natl. Acad. Sci. U. S. A., v. 112, no. 47, p. 14518–14521.
- Brent Dalrymple, G., 1994, The Age of the Earth: Stanford University Press.
- Burleigh, R., 1981, W. F. Libby and the development of radiocarbon dating: Antiquity, v. 55, no. 214, p. 96–98.
- Christopher B. DuRoss, Stephen F. Personius, Anthony J. Crone, Susan S. Olig, and William R. Lund, 2011, Integration of Paleoseismic Data from Multiple Sites to Develop an Objective Earthquake Chronology: Application to the Weber Segment of the Wasatch Fault Zone, Utah: Bulletin of the Seismological Society of America, v. 101, no. 6, p. 2765–2781., doi: 0.1785/0120110102.
- Dass, C., 2007, Basics of mass spectrometry, in Fundamentals of Contemporary Mass Spectrometry: John Wiley & Sons, Inc., p. 1–14.
- Elston, D.P., Billingsley, G.H., and Young, R.A., 1989, Geology of Grand Canyon, Northern Arizona (with Colorado River Guides): Lees Ferry to Pierce Ferry, Arizona: Amer Geophysical Union.
- Erickson, J., Coates, D.R., and Erickson, H.P., 2014, An introduction to fossils and minerals: seeking clues to the Earth’s past: Facts on File science library, Facts On File, Incorporated, Facts on File science library.
- Geyh, M.A., and Schleicher, H., 1990, Absolute Age Determination: Physical and Chemical Dating Methods and Their Application, 503 pp: Spring-er-Verlag, New York.
- Ireland, T., 1999, New tools for isotopic analysis: Science, v. 286, no. 5448, p. 2289–2290.
- Jackson, P.W., and of London, G.S., 2007, Four Centuries of Geological Travel: The Search for Knowledge on Foot, Bicycle, Sledge and Camel: Geological Society special publication, Geological Society, Geological Society special publication.
- Jaffey, A.H., Flynn, K.F., Glendenin, L.E., Bentley, W.C., and others, 1971, Precision measurement of half-lives and specific activities of U 235 and U 238: Phys. Rev. C Nucl. Phys.
- Léost, I., Féraud, G., Blanc-Valleron, M.M., and Rouchy, J.M., 2001, First absolute dating of Miocene Langbeinite evaporites by 40Ar/39Ar laser step-heating:[K2Mg2 (SO4) 3] Stebnyk Mine (Carpathian Foredeep Basin): Geophys. Res. Lett., v. 28, no. 23, p. 4347–4350.
- Mosher, L.C., 1968, Triassic conodonts from Western North America and Europe and Their Correlation: J. Paleontol., v. 42, no. 4, p. 895–946.
- Oberthür, T., Davis, D.W., Blenkinsop, T.G., and Höhndorf, A., 2002, Precise U–Pb mineral ages, Rb–Sr and Sm–Nd systematics for the Great Dyke, Zimbabwe—constraints on late Archean events in the Zimbabwe craton and Limpopo belt: Precambrian Res., v. 113, no. 3–4, p. 293–305.
- Patterson, C., 1956, Age of meteorites and the earth: Geochim. Cosmochim. Acta, v. 10, no. 4, p. 230–237.
- Schweitzer, M.H., Wittmeyer, J.L., Horner, J.R., and Toporski, J.K., 2005, Soft-tissue vessels and cellular preservation in Tyrannosaurus rex: Science, v. 307, no. 5717, p. 1952–1955.
- Valley, J.W., Peck, W.H., King, E.M., and Wilde, S.A., 2002, A cool early Earth: Geology, v. 30, no. 4, p. 351–354.
- Whewell, W., 1837, History of the Inductive Sciences: From the Earliest to the Present Times: J.W. Parker, 492 p.
- Wilde, S.A., Valley, J.W., Peck, W.H., and Graham, C.M., 2001, Evidence from detrital zircons for the existence of continental crust and oceans on the Earth 4.4 Gyr ago: Nature, v. 409, no. 6817, p. 175–178.
- Winchester, S., 2009, The Map That Changed the World: William Smith and the Birth of Modern Geology: HarperCollins.
![By Inglesenargentina (talk) (Uploads) (Own work) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3ABell-shaped-curve.JPG">via Wikimedia Commons</a> Mosquito trapped in amber in actal preservation](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.19_mosquito_in_amber-300x206.jpg)
![By User:Moondigger (Own work) [<a href="http://creativecommons.org/licenses/by-sa/2.5">CC BY-SA 2.5</a>], <a href="https://commons.wikimedia.org/wiki/File%3APetrified_forest_log_2_md.jpg">via Wikimedia Commons</a> Photo of log of petrified wood showing structures of the original wood](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.20_Petrified_forest_log_2_md-300x300.jpg)
![By Wilson44691 (Own work) [Public domain], <a href="https://commons.wikimedia.org/wiki/File%3AViburnumFossil.jpg">via Wikimedia Commons</a> carbonized leaf fossil showing insect damage and vein structure](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.23_carbonization_of_ViburnumFossil-300x215.jpg)
![<a href="https://en.wikipedia.org/wiki/User:Ballista" class="extiw" title="en:User:Ballista">Ballista</a> at the <a href="https://en.wikipedia.org/wiki/" class="extiw" title="w:">English language Wikipedia</a> [<a href="http://www.gnu.org/copyleft/fdl.html">GFDL</a> or <a href="http://creativecommons.org/licenses/by-sa/3.0/">CC-BY-SA-3.0</a>], <a href="https://commons.wikimedia.org/wiki/File%3ACheirotherium_prints_possibly_Ticinosuchus.JPG">via Wikimedia Commons</a> Tracks of an ancient 5-toed animal](https://opengeology.org/textbook/wp-content/uploads/2016/07/07.24_Cheirotherium_prints_possibly_Ticinosuchus-189x300.jpg)