A major innovation in living organisms came about through the cooperation of two early simple cells, working together for their common benefit. Endosymbiosis is a variety of mutual symbiosis in which one organism lives inside another. This process represents a merger of branches of the “tree of life,” and is thus a form of horizontal gene transfer. The evolution of organelles (small, functional units within cells) resulting from endosymbiosis have led to major revolutions in the history of life, including the first protists and the eventual development of multicellular organisms such as plants, fungi, and animals.
Making eukaryotes from prokaryotes
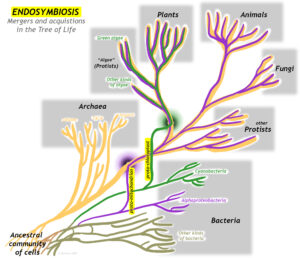
Eukaryotic cells are the large, complicated cells that make up the bodies of animals, plants, fungi, and protists. With their well-defined nucleus, they are distinct from the smaller, simpler cells that comprise the bodies of single-celled microbes. Microbial cells are prokaryotes instead, and come in two varieties: bacteria and archaea. Bacteria and Archaea strongly resemble one another in size and form (including their lack of a nucleus), but are distinguished from one another on the basis of utterly distinct genetics and biochemistry.
Though the domain Eukarya contains a diversity of forms, the defining characteristic of a eukaryote has long been recognized as its membrane-bound nucleus, the site where its genetic material is stored (in the form of DNA) and transcribed (in the form of RNA). The –karyotic part of the names “eukaryote” and “prokaryote” translates as kernel or nugget, a reference to the distinct visibility of the nucleus in those cells that have one. However, the mitochondrion could be considered just as defining and ubiquitous a structure for eukaryotes. Only a few anaerobic eukaryotes lack mitochondria. The structure, function, and genetic signature of the mitochondrion gives every indication that it represents the “capture” of a bacterial cell by an archeal cell sometime around 1.45 Ga (Mesoproterozoic).
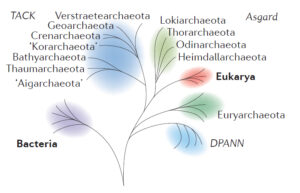
Modified from Figure 1 of Eme, et al. (2017).
There is great diversity among the Archaea, and several major groups within that domain had split off already by the time one sub-group had some members engage in endosymbiosis while others didn’t. The group of Archaea most closely related to the eukaryotes are the superphylum Asgard. The Asgardians are more related to the “Eukaryotic” host cell line than they are to other groups classified as fellow archeons. Somewhere at or just after the branching point between Asgard and Eukarya was the moment when the first proto-mitochondrion was engulfed. The endosymbiosis between the Asgard-like archeon and its future mitochondrion became cemented into a permanent and irrevocable interdependency through geologic time. What was initially perhaps happenstance and casual eventually evolved into something permanent and obligatory, an innovation that was wildly successful in allowing diversification of life on Earth.
Endosymbiosis is often thought of as the larger organism “engulfing” the smaller, but it could just as legitimately be viewed as the “colonization” of the larger organism by the smaller one. Either way, each organism benefits from the relationship. The net result is a genetic chimera, a blending and a merger between two organisms that used to be distinct, but when combined make something new and different. All eukaryotes are chimeras, some several times over.
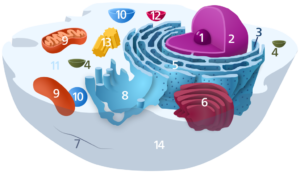
1) Nucleolus; 2) Nucleus; 3) Ribosome (dots as part of 5); 4) Vesicle; 5) Rough endoplasmic reticulum; 6) Golgi apparatus; 7) Cytoskeleton; 8) Smooth endoplasmic reticulum; 9) Mitochondrion; 10) Vacuole; 11) Cytosol (fluid that contains organelles; with which, comprises cytoplasm); 12) Lysosome; 13) Centrosome; 14) Cell membrane.
Public domain image via Wikimedia.
What is true for mitochondria among all eukaryotes is also true for another organelle in some eukaryotes: plants and the various forms of “algae” (which are protists) contain chloroplasts, organelles that have the ability to conduct photosynthesis. In structure, function, and genes, the chloroplasts resemble free-living cyanobacteria, and are thus thought to represent a second episode of endosymbiosis unique to the photosynthetic eukaryotes.
In both cases, the smaller symbiont retains its own DNA, and reproduces independently of the rest of the host cell. Over geologic time, the genome of the internal symbiont will be pared down to eliminate redundancy, with the genome sometimes getting trimmed down to a third the size of the length of the DNA in their free-living relatives. Some of its genes may be copied or moved into the host genome. The two organisms may become locked into interdependency, where neither can survive without the other. At this point, it starts to make more sense to refer to the internal symbiont not as an “organism” in its own right, but as an organelle within the host cell.
There are two major organelles in eukaryotic organisms that have resulted from endosymbiosis, mitochondria (common to most eukaryotes) and chloroplasts (found in plants and algae only), as well as a few less obvious examples. After reviewing the scientific history of the idea, this case study will examine each organelle in turn.
Did I get it?
A history of the idea
In the early 1880s, German botanist Andreas Schimper used the most cutting-edge microscopes available to study plant cells. He noted that chloroplasts grew and divided independently of the rest of the cell. This was a novel insight, and confounded expectations. Copying the experiments of his contemporary Louis Pasteur, Schimper demonstrated that a plant cell which has lost its chloroplasts cannot regenerate them. Like life itself, in the modern day, all chloroplasts descend from other chloroplasts. Schimper’s 1883 conclusion was that chloroplasts were endosymbiotic cyanobacteria. Another botanist, this one from Russia, Konstantin Merezhkovsky (also spelled Mereschkowski) built on this idea, proposing in 1905 that algae and plant cells were chimeras between a protozoan (a protist) and a cyanobacterium nestled inside it. However, critical empirical tests were missing at the turn of the century: there was no DNA sequencing available then, nor sufficiently advanced microscopy to investigate Merezhovsky’s ideas to the extent they deserved. Thus, the hypothesis of chloroplast endosymbiosis withered shortly after it was first suggested. The technology that would show it to be true had not yet been invented. An idea this radical needed testing to remain viable; Without it, the idea went dormant for several decades.
Shortly thereafter, American biologist Ivan Wallin developed an interest in mitochondria. In 1922, he noted that the little rod-shaped organelles were so similar to bacteria that the two were frequently mistaken for one another. Intriguingly, the same staining techniques that worked to highlight mitochondria also worked on bacteria. Wallin concluded that, “mitochondria are symbiotic bacteria in the cytoplasm of the cells of all higher organisms.” Again, the idea was plainly stated but did not gain widespread acceptance due to the lack of confirmation from other more precise lines of evidence.
At the same time as Wallin’s work in America, another botanist in Russia independently came up with the same idea expressed decades earlier by Merezhovsky. In 1924, Boris Kozo-Polyansky published a paper again documenting the similarities between chloroplasts and cyanobacteria, extrapolating the larger insight that, “new organisms occur not only by divergence of the lineages but also by their convergence and fusion.” Again, the ground was not fertile for the reception of the provocative notion, and it once more faded into obscurity.

Photo by J. Pedreira via Wikimedia Commons.
The idea of endosymbiosis would see a revival and ultimate validation in 1967 when Lynn Margulis (then Lynn Sagan) [yes, that Sagan] proposed that not only was the endosymbiosis notion true for chloroplasts, it also applied to mitochondria, and maybe flagella too. A rhetorical force of nature, when Margulis championed endosymbiosis, it stuck. Widespread scientific consensus wasn’t only the result of Margulis’s tireless advocacy, but also because of the advance in scientific technology since Merezhovsky’s day. For one thing, electron microscopy now existed, and was capable of showing how similar the internal structure of chloroplasts was to the architecture of cyanobacteria. Moreover, it showed the double-membraned envelope of chloroplasts, a signature of having been enfolded from the exterior to the interior of the host eukaryote. Seeing these details simply wasn’t possible prior to sufficiently detailed imaging technology.
More recently, the ability to extract and sequence DNA has shown the similarity between the genes of organelles like the mitochondrion and the chloroplast and free-living bacteria. The two structures shared many of the same genes, in the same way that familial cousins do. This was the detail that cinched the case for most scientists. With multiple lines of evidence converging on the same answer, it was time to accept that answer: Eukaryotes are chimeras resulting from endosymbiosis.
Because eukaryotes are capable of feats that could never be accomplished by a lone prokaryote, a philosophical corollary is that evolutionary innovation can be achieved by cooperation rather than solely through competition. The idea of endosymbiosis offers a different lens through which to view the evolution of life on Earth: it turns “survival of the fittest” on its head, and suggests instead “survival of the most well-integrated team.”
Did I get it?
Mitochondria
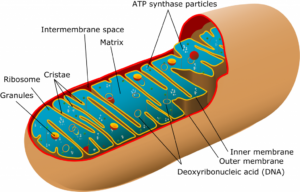
Illustration by Mariana Ruiz Villarreal, via Wikimedia Commons.
Mitochondria are organelles found in many eukaryotes, including animals, plants, fungi, and many varieties of protist. They are bounded by two membranes, the outer one smooth and pill-shaped, the inner one intensely folded into fin-like creases. This gives the inner membrane a very large surface area, crumpled into a small volume of space. On average, they are about 1 μm long. The number of mitochondria per cell can vary between zero (in some anaerobic protists) and upward of 2000. Functionally, mitochondria are critical because they produce the molecule adenosine triphosphate (ATP), a vital molecule used as a conveyance of chemical energy in the cell. To do this, they catalyze the reaction of free oxygen (O2) with pyruvate (a product of the digestion of glucose), and generate carbon dioxide (CO2) and water (H2O) as waste products in addition to the ATP. In most mitochondria in most eukaryotes, about 36 units of ATP are produced as outputs per unit of glucose ingested. In other words, mitochondria are where respiration happens: where energy is derived from the food we eat, “burning” it chemically by oxidizing it, and “charging up the batteries” of our bodies.
Mitochondria are probably descended from a common ancestor shared with modern free-living bacteria, specifically the group called alphaproteobacteria (sometimes written as α-proteobacteria). We think this for several reasons. First, mitochondria only come from other mitochondria. Like bacteria, they reproduce through binary fission. If they are removed from a cell, the cell cannot replace them on its own. Within the inner membrane, the mitochondrion contains DNA, RNA, and ribosomes: all the apparatus for storing genetic information and translating it into proteins. Second, comparison of the ribosomal RNA in mitochondria and other extant groups shows the mitochondria to be most similar to the alphaproteobacteria. The extant alphaproteobacterial genus Rickettsia, a tickborne pathogen, comes closest in relatedness to the proto-mitochondrion. Interestingly, the tetracycline antibiotic medications used to treat tickborne illnesses such as Rocky Mountain spotted fever (such as Doxycycline) can also negatively impact the functioning of mitochondria in animal cell test cultures.
Did I get it?
Recent analogues
The modern amoeba species Pelomyxa hosts endosymbiotic bacteria that serve the same function as mitochondria do in other eukaryotes. Interestingly, ribosomal RNA analysis of its endosymbionts reveals not one species but three! Moreover, two of them are bacterial, while one is a methanogenic archeon (Gutiérrez et al., 2017). This set of relationships is probably relatively recent, for it is not sufficiently entrenched as to be obligatory. The microbial symbionts can live freely on their own, and Pelomyxa has not yet evolved a system of passing them to its descendants. So the amoeba must capture fresh bacterial endosymbionts anew with each generation.
Strigomonas is another example. This trypanosomatid is related to the parasite that causes sleeping sickness in humans. It relies on an endosymbiotic betaproteobacterium, and cannot live without it, though the bacteria is capable of life outside Strigomonas. This is an interesting case where one relies on the other, but the feeling is not mutual!
Mitochondrial DNA
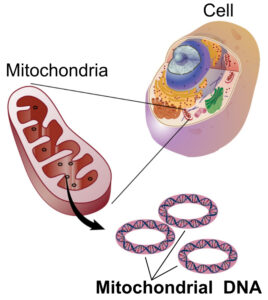
Illustration by National Human Genome Research Institute – National Institutes of Health. (Public domain)
The DNA of mitochondria is not stored in chromosomes, the way that nuclear DNA is. Instead, it is found in circular “hoops” of DNA within the mitochondrion, similar to the shape in which DNA is organized in bacteria. In animal mitochondria, the length of a one of these hoops ranges from 11–28 thousand base pairs. (For comparison, the number of base pairs in the human genome is ~3.2 billion.)
In animals, mitochondrial DNA is passed from mother to offspring. The father contributes half of the nuclear genetic material to his children, but the mother supplies the other half of the nuclear DNA plus all of the mitochondrial DNA. You got your mitochondria from your mom, who got them from her mom (your maternal grandmother), who got them from her mom (your mom’s maternal great-grandmother). Mitochondrial DNA is thus useful for tracing a line of matrilineal descent.
Studies of human genetics has shown a mitochondrial last common ancestor to all mitochondria in living humans was in a woman who lived in Africa between 120,000 and 156,000 years ago. This individual has been dubbed “mitochondrial Eve.” [LINK TO HUMAN EVOLUTION CASE STUDY?]
Chloroplasts
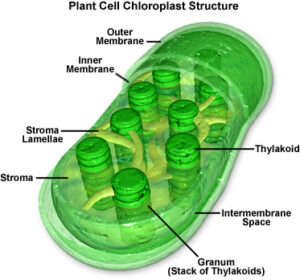
Image by MolecularExpressions at Florida State University; reproduced with permission.
Chloroplasts are organelles within the cells of algae and plants that are the sites of the photosynthesis reaction. There can be between one and ~100 chloroplasts per plant cell. They are less than 10 μm in diameter and ~2 μm thick. Within each chloroplast are multiple stacks of poker-chip-shaped discs called thylakoids. These structures contain chlorophyll, the enzyme which utilizes incoming wavelengths of light in the blue and red portions of the visible spectrum to catalyze the reduction of atmospheric carbon dioxide (CO2) with water (H2O), generating glucose (C6H12O6), along with free oxygen (O2) as a waste product. (Chlorophyll appears green because that is the portion of the spectrum not utilized for photosynthesis. Rather than being absorbed, it is reflected.)
The anatomy of chloroplasts is shared by free-living cyanobacteria. Like cyanobacteria, chloroplasts have their own DNA. They reproduce independently of the rest of their host cell. Unlike cyanobacteria, they have two membranes separating them from their host cell. They make the inner of the two membranes, while the outer membrane is manufactured by the host cell.
We estimate that the endosymbiosis between an early eukaryote (already sporting mitochondria) and a cyanobacterium (the proto-chloroplast) to have occurred around 1.25 Ga. This date is supported both by genetic comparisons and the fossil record. It probably happened more than once, something that is thought to be more likely on the extreme population sizes and ubiquity of bacteria.
Did I get it?
Recent analogues
Photograph by Ferry J. Siemensma; reproduced here with permission.
An illuminating analogue can be found in the amoeba Paulinella, which incorporated a cyanobacterium into its cells much more recently, sometime between 140 and 90 Ma. Based on genetic similarity, the internal symbiont was probably an ancestor to one of the extant marine cyanobacterial genera Prochlorococcus or Synechococcus. Though the symbiosis that led to proper chloroplasts is more obscured by the depths of geologic time, that the same process has been “done over” independently in other groups such as Paulinella suggests the validity of the idea of cyanobacterial endosymbiosis as a critical process in the evolution of photosynthetic eukaryotic life.
Another example is Azolla. Technically a fern, it looks nothing like most ferns, and instead floats on the surface of freshwater ponds all over the world. Azolla hosts the cyanobacterium Anabaena within its cells. The Anabaena has the ability to capture nitrogen (N2) from the surroundings and “fix” it in a biologically useful form, ammonia (NH3). This is beyond the abilities of a typical chloroplast, but being a plant the Azolla already has typical chloroplasts. The endosymbiosis of the Anabaena is an additional cyanobacterial acquisition, for the sake of the nitrogen it can sequester and share. This is a huge advantage to the Azolla host, which is extremely capable of growth as a result – it can double its biomass in two days. The relationship between Azolla and Anabaena is not haphazard or improvised: It’s codified enough that the host Azolla cells pass the endosymbiont Anabaena cells directly to their offspring through cell division and reproduction, an example of “vertical” transmission.
Take two
There can also be multiple rounds of endosymbiosis. Imagine, for instance, a eukaryotic cell containing a double-membraned chloroplast. (This is the situation we have already described.) What happens when that cell gets engulfed by another, bigger cell? This is secondary endosymbiosis, a fresh iteration wherein the eater gets eaten. The chloroplast will now be quadruple membraned, with both the “original” two membranes, plus a third membrane from its first host and a fourth membrane from its newest, outermost host.
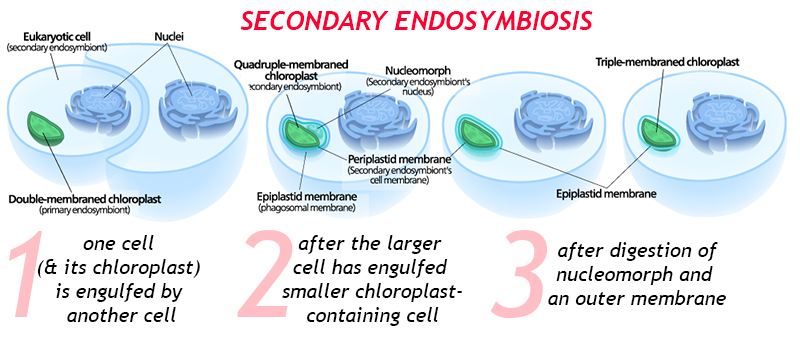
When this process is recent, there may be a residual nucleus-like body between the third and fourth membranes: this is the nucleus of the first host cell. In time, the second host cell may digest (or assimilate) that DNA, and perhaps one or more of the membranes surrounding the chloroplast.
There is even a well-documented case of tertiary endosymbiosis, in the dinoflagellates, where a cyanobacterium was engulfed by an ancestral eukaryotic host, making a unicellular alga, which was then swallowed by a protozoan, and then that was engulfed by a haptophyte alga! Geobiologist Andy Knoll refers to dinoflagellates as “biological Matryoshka dolls,” for good reason. A good first guess about how many episodes of endosymbiosis a particular organelle has undergone can be arrived at by counting the number of membranes and subtracting 1. Just to drive the point home, it’s worth noting that dinoflagellates are the so-called “zooxanthellae” that are endosymbionts in corals: Unless the coral undergoes “bleaching,” evicting the dinoflagellates into the surrounding water, that’s one more Matryoshka doll yet!
Did I get it?
Other organelles
Though mitochondria and chloroplasts are the most well known and important examples of microbial endosymbiosis leading to novel organelles, there are perhaps other examples too.
Consider the flagellum, a whip-like appendage that projects off the surface of many groups of microbes. Lynn Margulis suggested that the flagellum appears to be an organelle that represents a merger between a spirochete bacterium and a host archeal cell related to the genus Thermoplasma. This idea hasn’t been as thoroughly validated as the endosymbiotic origins of mitochondria and chloroplasts, but it remains an intriguing idea worth further investigation.
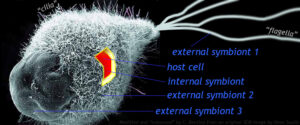
Modified from an SEM image by Dean Soulia.
Mixotricha paradoxa is an interesting example that appears to corroborate the idea of a symbiotic origin for flagella. Mixotricha is a species of protozoan (a eukaryote) that lives inside the gut of certain Australian termites. Unusually for protozoans, it sports both cilia and flagella, which is why its name translates to “a paradoxical mixture of hairs.” Mixotricha has five genomes: one for the main host cell, one for an endosymbiont that helps with the digestion of cellulose and lignin, and three on the surface (two spirochetes and one rod bacterium).
Some scientists have suggested that the nucleus itself, that iconic hallmark of the eukaryotic cell, arose through an even earlier episode of endosymbiosis. Perhaps that earliest, most fundamental merger occurred with a now-extinct group of microbes. Hartman and Federov (2002) identified hundreds of genes coding for proteins that are found in eukaryotic cells but have no equivalents in either Archaea or Bacteria. They interpret this as evidence of a third microbial line, the primordial host cell that would eventually lead to the eukaryotic line, dubbing it the Chronocyte (translation = time cell). Another possibility is that a large DNA virus may have been responsible; this is the viral eukaryogenesis hypothesis. Future research may shed more light on these intriguing possibilities.
An intriguing series of mergers and acquisitions permeate the history of eukaryotes on Earth, some more plain and others more obscure. When we consider ourselves as individuals, its humbling to realize we are in fact communities within communities, a whole that is greater than the sum of its parts.
Did I get it?
Further reading
Iliana Chatzispyrou, Ntsiki Held, Laurent Mouchiroud, Johan Auwerx, & Riekelt Houtkooper (2015). “Tetracycline antibiotics impair mitochondrial function and its experimental use confounds research,” Cancer Research vol. 75, 21: pages 4446-9. doi:10.1158/0008-5472.
Delaye L, Valadez-Cano C, Pérez-Zamorano B. (2016). “How Really Ancient Is Paulinella Chromatophora?” PLOS Currents Tree of Life, 2016 Mar 15 . Edition 1. doi: 10.1371/currents.tol.e68a099364bb1a1e129a17b4e06b0c6b.
Laura Eme, Anja Spang, Jonathan Lombard, Courtney Stairs, & Thijs Ettema. (2017). “Archaea and the origin of eukaryotes,” Nature Reviews Microbiology 15 (12):711-723; 2017 Nov 10. PMID: 29176585.
Hyman Hartman & Alexei Fedorov (2002). “The origin of the eukaryotic cell: A genomic investigation,” Proceedings of the National Academy of Sciences, Feb 2002, 99 (3) 1420-1425; DOI:10.1073/pnas.032658599
Andy Knoll (2004). Life on a Young Planet. Princeton University Press, 288 pages.
William F. Martin & Marek Mentel (2010). “The Origin of Mitochondria,” Nature Education 3 (9):58.
Oliver Morton (2008). Eating the Sun. Harper Collins, 445 pages.
PBS Digital Studios (2018). “How two microbes changed history,” Eons television program, 7 minutes & 39 seconds.
Lynn Sagan [Margulis] (1967). “On the origin of mitosing cells,” Journal of Theoretical Biology 14 (3), pages 225-IN6, ISSN 0022-5193, https://doi.org/10.1016/0022-5193(67)90079-3.
Carl Woese & George Fox (1977). “Phylogenetic structure of the prokaryotic domain: the primary kingdoms,” Proceedings of the National Academy of Science of the United States of America 74, 5088–5090.